FDA inspection found problems at factory making J&J vaccine
U.S. regulators say the Baltimore factory contracted to make Johnson & Johnson’s COVID-19 vaccine was dirty, didn’t follow proper manufacturing procedures and had poorly trained staff
Your support helps us to tell the story
From reproductive rights to climate change to Big Tech, The Independent is on the ground when the story is developing. Whether it's investigating the financials of Elon Musk's pro-Trump PAC or producing our latest documentary, 'The A Word', which shines a light on the American women fighting for reproductive rights, we know how important it is to parse out the facts from the messaging.
At such a critical moment in US history, we need reporters on the ground. Your donation allows us to keep sending journalists to speak to both sides of the story.
The Independent is trusted by Americans across the entire political spectrum. And unlike many other quality news outlets, we choose not to lock Americans out of our reporting and analysis with paywalls. We believe quality journalism should be available to everyone, paid for by those who can afford it.
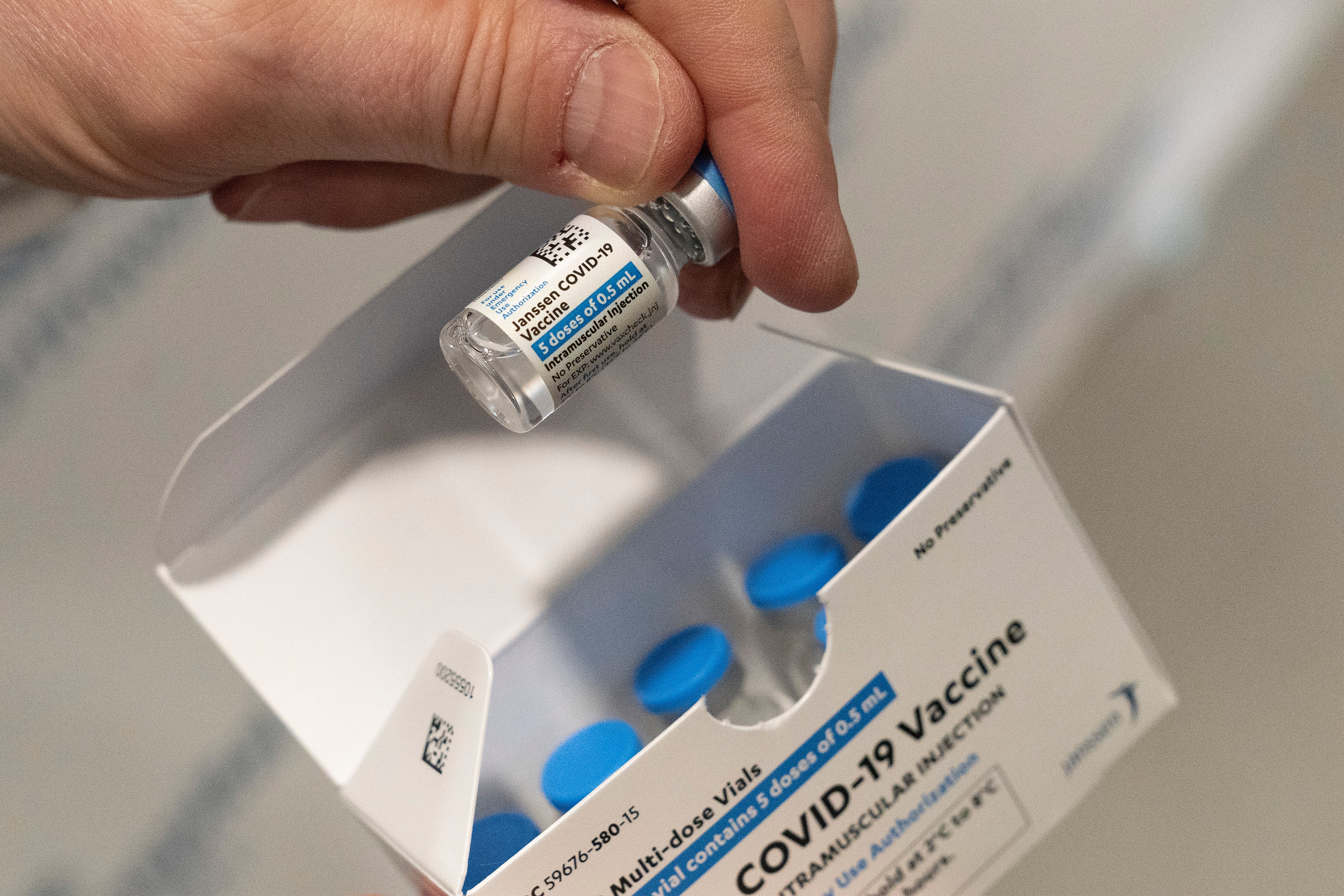
Your support makes all the difference.The Baltimore factory contracted to make Johnson & Johnson’s COVID-19 vaccine was dirty, didn’t follow proper manufacturing procedures and had poorly trained staff, resulting in contamination of material that was going to be put in the shots, U.S. regulators said Wednesday.
The Food and Drug Administration released a statement and a 13-page report detailing findings from its recent inspection of the now-idle Emergent Biosciences factory.
Agency inspectors said a batch of the bulk drug substance for J&J's single-shot vaccine was contaminated with material used to make COVID-19 vaccines for another Emergent client, AstraZeneca That batch, reportedly enough to make about 15 million J&J vaccine doses, had to be thrown out.
Other problems cited in the inspection report were peeling paint, black and brown residue on floors and walls in the factory, inadequate cleaning and employees not following procedures to prevent contamination.
Both Emergent and Johnson & Johnson said Wednesday that they are working to fix the problems as quickly as possible. Nothing made at the factory for J&J has been distributed yet.
At the moment, use of the J&J vaccine is on hold in the U.S. as government health officials investigate its connection to very rare blood clots. The nearly 8 million doses of J&J vaccine that had been used in the U.S. came from European sources.
The Baltimore factory halted production late last week at the request of the FDA The agency hasn’t given emergency approval to the factory, which is needed before any vaccine material made at the factory can be distributed.
All the bulk vaccine substance Emergent has made is being stored and will undergo additional testing by the FDA, the agency said.
___
The Associated Press Health and Science Department receives support from the Howard Hughes Medical Institute’s Department of Science Education. The AP is solely responsible for all content.